PEGYLATED LIPOSOMES
In order to enable intracellular delivery of drugs and medicines, a vehicle molecule is required. Such vehicle molecules vary in type, mechanism of action, targeting of specific organs or tissues, and unique physical and chemical properties. PEG-liposomal delivery technology is known to be superior in terms of delivery efficiency and tissue targeting applications while inducing minimal immune response. Preclinical contract research organizations (such as Altogen Labs) provide PEG liposome encapsulation services (see service description here) for in vivo RNAi and multiple other gene expression in vivo applications. Encapsulated mRNA, siRNA, shRNA, plasmid DNA, proteins and small molecules are used for both in vitro and in vivo transfection laboratory experiments.
Cationic polymers that form stable positively charged nanoparticles with DNA in water are commercially available for in vivo transfection. Cationic lipids are commonly used to deliver nucleic acids due to electrostatic interactions with amphiphilic molecules. There are many methods of introducing nucleic acids into an organism usually referred to as gene therapy. Some of the more successful ones include:
- • Injection of naked DNA – mixed success, often cleared without effect
- • Sonoporation method
- • Electroporation method
- • Lipoplexes method
- • Polyethylenimine (PEI) reagent can be used to deliver genes or RNA via a spinal catheter in vivo for successful transfection.
PEG (POLYETHYLENE GLYCOL) TRANSFECTION COMPLEXES
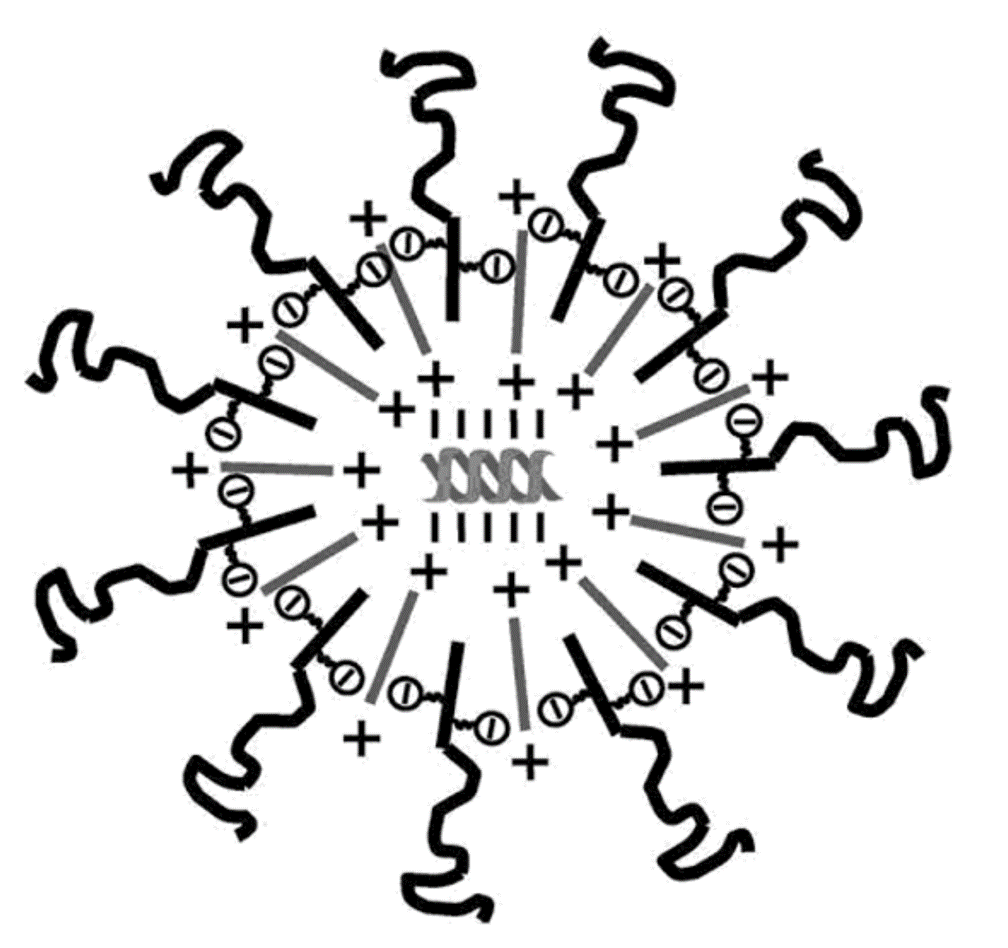
A liposome is a very simple cellular structure that can carry other molecules inside the cell, including small drugs or a segment of genetic code (DNA or RNA). These fundamental structures are found abundantly in and around cells and can be artificially created. Historically, liposome mediated delivery has been utilized for many decades. However, in vivo liposome delivery systems experience immediate elimination from circulation. Positively charged complexes interact with negatively charged components in plasma, with liposome binding to lipoproteins resulting in inactivating the complexes. The lipoprotein interaction with lipoplexes forms a bond between the two components that leads to aggregate formation.
To circumvent elimination and aggregation, PEG-lipids can be inserted into the bilayer of the lipid complex. The addition of the PEG-lipids shields the lipid complex from being cleared from circulation to increase bioavailability and limits aggregation. PEG-lipids are commonly used in pharmaceuticals due to its ability to stabilize liposomes and low molecule cost. Formulation chemists routinely alter the PEG chain length, PEG density, spacers and linkers to customize zeta-potential, complex size and plasma interaction.
PEG-liposomes are liposomal complexes (cargo molecules include plasmid DNA, mRNA, small RNA, small molecules, and proteins) that do not induce immune response and provides superior in vivo delivery compared to many counterparts. PEG-liposomes are commercially available from Altogen Biosystems – PEG-Liposome Kits.
PEG IN VIVO TRANSFECTION PROTOCOL
PEGYLATED LIPOSOME DESCRIPTION
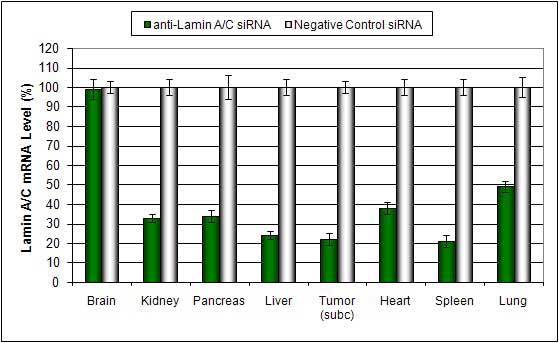
PEGylated cationic lipid liposome-based In Vivo Transfection Reagent is a lipid-functionalized PEG complex proprietary formulation optimized for in vivo delivery of miRNA, siRNA, plasmid DNA and proteins. PEG-Liposome complexes are stable for at least 16 hours in serum. There is no detectable inflammatory response or toxicity due to the PEG modification. Efficient delivery has been exhibited to these tissues: pancreas, spleen, kidney, liver and tumors via systemic administration.
PEGYLATED LIPOSOME SYSTEMIC ADMINISTRATION (I.V. INJECTION)
Below is a recommended protocol for the intravenous injection of PEGylated liposomes for in vivo experiments.
- Dilute 60 µg of plasmid DNA or 100 µg of siRNA in 100 µL nuclease-free water and vortex gently.
- Add 100 µL of the diluted to a sterile tube containing 50 µL Transfection Reagent.
- Incubate for 15-20 min at room temperature.
- Add 10 µL of Transfection Enhancer Reagent and vortex gently to mix.
- Incubate for 5 min at room temperature.
- Add required amount of sterile solution of 5% glucose (w/v):
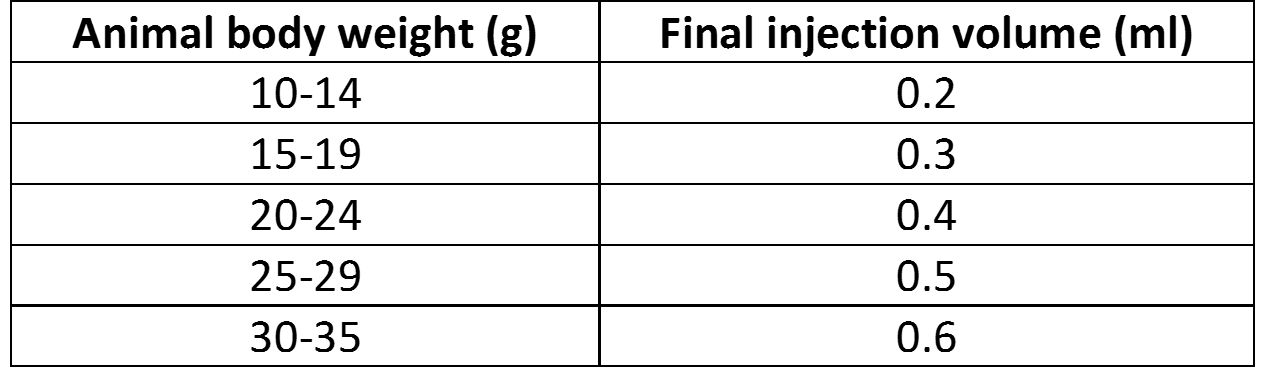
- Inject animals: Please note that delivery efficiency can be significantly increased by performing a secondary injection (at least 12 hours after first injection).
- Maximum mRNA target expression effect is typically observed 12-36 hours after injection, while maximum effect on protein level is achieved 24-48 hours post-injection.